Tuberous sclerosis complex (TSC) is a rare disease. Only 1 in 6,000 children born in the United States may have TSC. The disease causes benign (noncancerous) tumors to grow throughout the body. These tumors can develop in many organs, including the brain, eyes, kidneys, liver, lungs, heart, and skin.
TSC is a genetic (inherited) disorder. It is caused by defects in 2 genes called TSC1 and TSC2. The symptoms and severity of TSC may vary widely—even among members of the same family. The tumors can grow in different places, in different numbers, and in different sizes. TSC can be so mild that some people are never diagnosed with it, or it can cause serious complications and even be life-threatening.
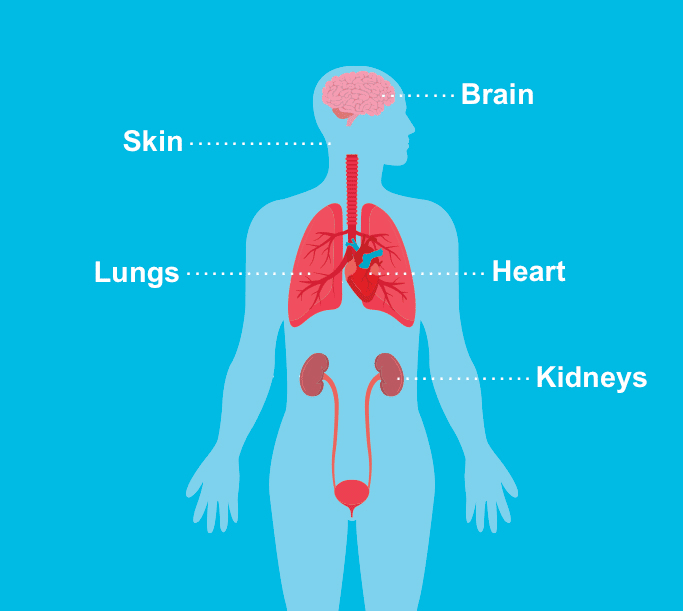
The skin is one of the most affected areas in people with TSC.
Facial angiofibromas are pinkish or reddish bumps that are usually located on the cheeks, nose, and chin. Although they can appear in other areas of the skin, facial angiofibromas often appear in a butterfly pattern across the nose and cheeks.
Facial angiofibromas are made up of blood vessels and tough tissue, and they can build up into larger patches. Although facial angiofibromas are benign, they can bleed, block the nasal openings, and cause disfigurement.
Facial angiofibroma occurs in approximately 75%-80% of TSC patients—making facial angiofibroma one of the most common skin conditions seen in TSC.
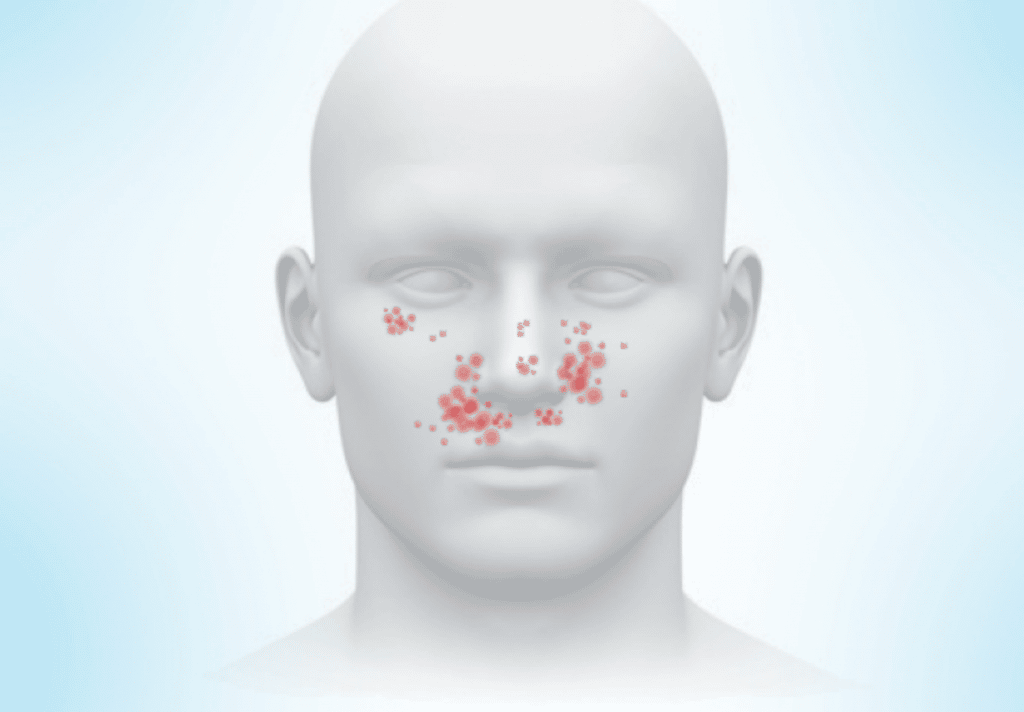
Facial angiofibromas can appear in early childhood. Then they can increase and grow larger as people age. At first, facial angiofibromas can cause a reddish or rosy appearance on the cheeks. However, they eventually become rougher and thicker.

Facial angiofibromas can cause disfigurement if left untreated. These tumors may also be a psychological and social burden for people living with this condition. Patients have reported that facial angiofibroma can have negative effects on appearance and self-image, causing some people to avoid social situations.
Before using HYFTOR®, tell your healthcare provider about all of your medical conditions, including if you:
What are possible side effects of HYFTOR®? HYFTOR® may cause serious side effects, including:
You are encouraged to report side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also reach out to Nobelpharma America, LLC at 1-877-375-0825
© 2022 Nobelpharma America, LLC. All rights reserved. HYF-PRO-1214-v3
What is HYFTOR®?
Link pending once FDA approval comes in…